JOINTS 2025;
3: e1599
DOI: 10.26355/joints_20259_1599
Arthroscopic reduction and internal fixation of anterior tibial spine: a practical guide
Topic: Knee
Category: Technical Note
Abstract
Tibial spine fractures, common among children and adolescents, involve the avulsion of the anterior cruciate ligament at its tibial insertion. Trauma is often the result of injuries occurring during high-intensity sports. Nondisplaced fractures can be treated conservatively, but more severe cases require surgical intervention to avoid long-term complications. Arthroscopic Reduction and Internal Fixation (ARIF) has gained favor due to its minimally invasive nature and the possibility of addressing associated soft tissue injuries.
This paper provides accurate information on the management of tibial spine avulsion fractures, including a detailed description of our ARIF technique, and its potential advantages and disadvantages over the open reduction internal fixation (ORIF) technique; postoperative outcomes are also discussed, and clinical cases derived from the authors’ experience are presented.
ARIF for tibial spine avulsion fractures, using a standardized approach with tailored execution, can provide optimal functional and radiological outcomes with minimal invasiveness, allowing a comprehensive treatment of associated intra-articular injuries.
Introduction
Tibial spine fractures, also known as tibial eminence fractures, represent an avulsion of the bony attachment of the anterior cruciate ligament (ACL) at the tibial insertion site. These fractures are more frequently observed in pediatric and adolescent populations and occur in 3 per 100,000 children annually, accounting for 2% to 5% of pediatric knee injuries with effusion. They affect males more frequently than females1. These fractures were historically associated with bicycle accidents, but the surge in popularity among children and adolescents of sports that involve high-intensity movements, such as football, skiing, or basketball, has brought a renewed interest in them.
The age span between 8 and 14 years is the most represented in this kind of injury. The reason could be plausibly linked to an incomplete ossification of the tibial spine, which can predispose young athletes to this type of fracture2. The mechanism of injury is often a combination of hyperextension and rotation of the knee or forced knee flexion with simultaneous uncontrolled tibial external rotation, mimicking the mechanism involved in ACL tears seen in adults.
The general clinical presentation consists of knee pain, swelling, and a reduced range of motion, often with the inability to fully extend the knee. In more severe cases, the presence of a palpable effusion may suggest intra-articular bleeding3.
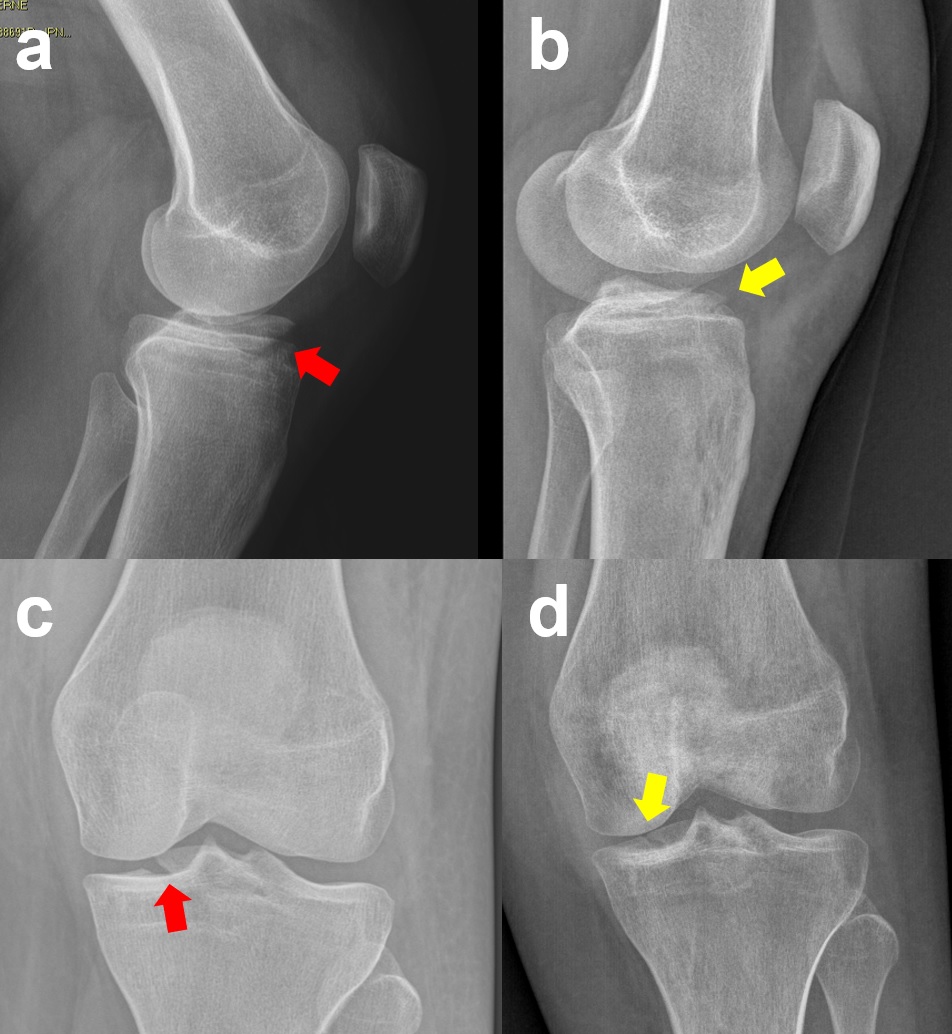
Conventional X-rays are often used as the initial diagnostic tool to identify fractures. CT scan is generally used in ambiguous cases and to better determine the degree of displacement4 (Figure 1). Based on that, the fracture can be classified according to the Meyers and McKeever’s classification in: Type I: minimally/nondisplaced fragment (< 3 mm); Type II: anterior elevation of the fragment; Type III: complete separation of the fragment (Type IIIa involves a small portion of the eminence; Type IIIb involves the majority of the eminence); Type 4: comminuted avulsion or rotation of the fracture fragment5. Alternatively, Green et al6 highlight the key role of MRI in the assessment of tibial eminence fracture because of the possibility to identify associated injuries such as meniscal tears (Figure 2) or ligamentous damage6,7. In our experience, additional soft tissue injuries such as meniscal entrapment or ACL injuries are identified alongside the fracture in a substantial number of cases8-10. The treatment approach depends on the severity of the fracture, with nondisplaced fractures often treated conservatively with immobilization in extension for 4-6 weeks11. However, in cases where there is a significant displacement, surgical intervention is typically required to prevent long-term instability, nonunion, or malunion12. Different surgical approaches have been proposed to manage tibial avulsion fractures. Among them, Arthroscopic Reduction and Internal Fixation (ARIF) has gained increasing preference over open techniques due to its minimally invasive nature, fast recovery times, and limited complications.
This study aims to present a detailed step-by-step description of our usual ARIF technique, which includes modifications of several existing techniques, and its differences from open reduction internal fixation (ORIF) surgery13-15.
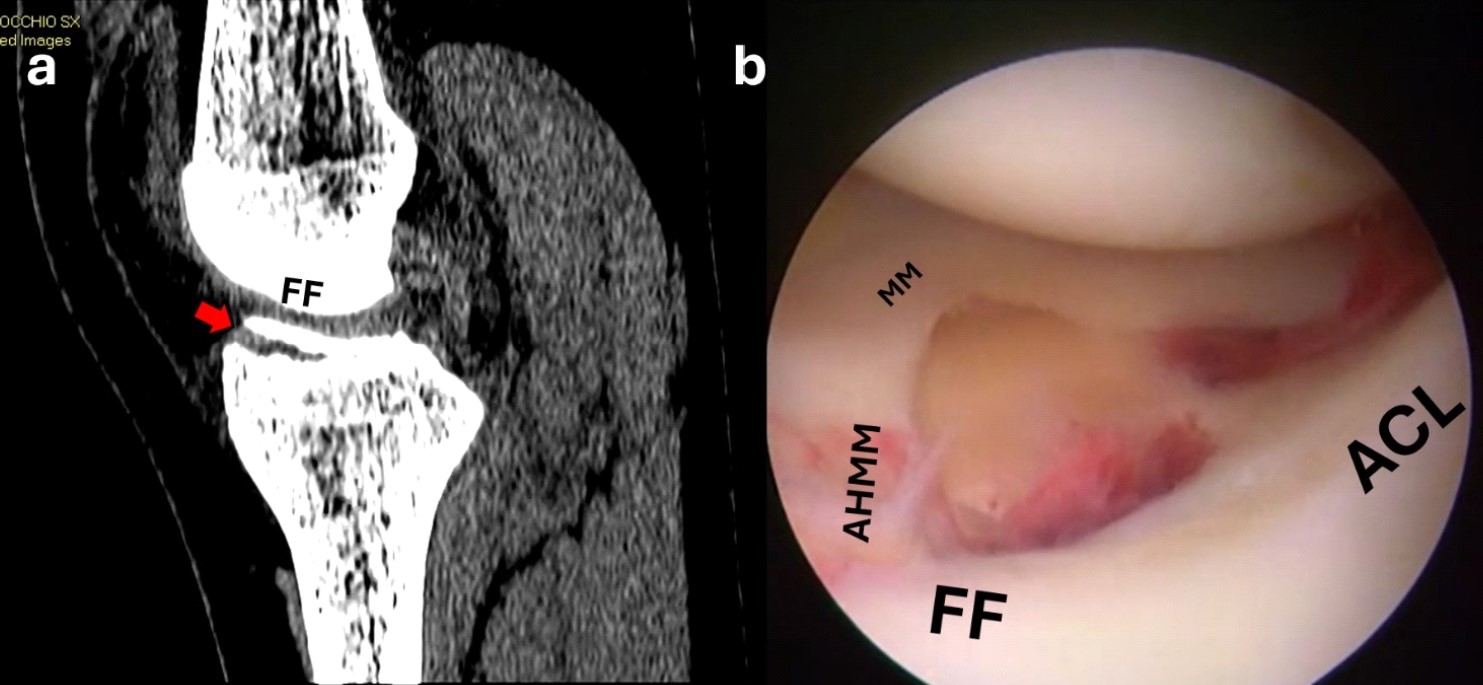
Figure 1. Sagittal CT scan showing a displaced anterior eminence fracture (red arrow) (a); the arthroscopic view shows the interposition of the intermeniscal ligament and the fracture bed (b). FF, fracture fragment; MM, medial meniscus; MFC, medial femoral condyle.
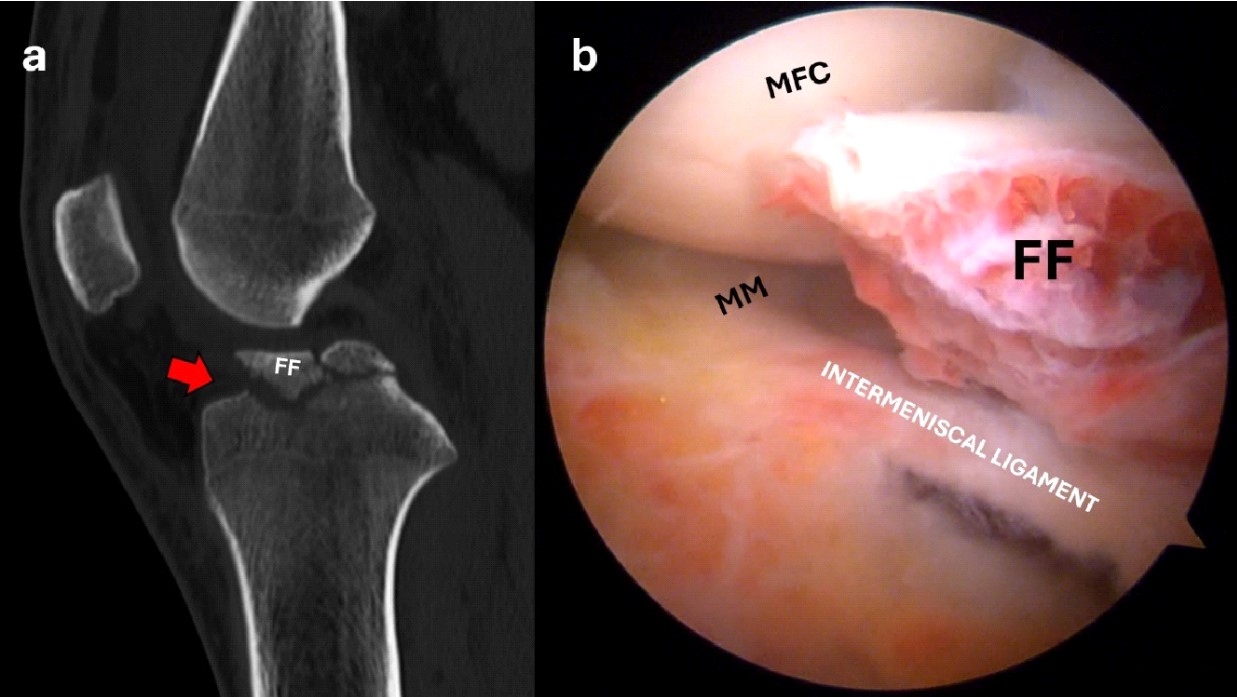
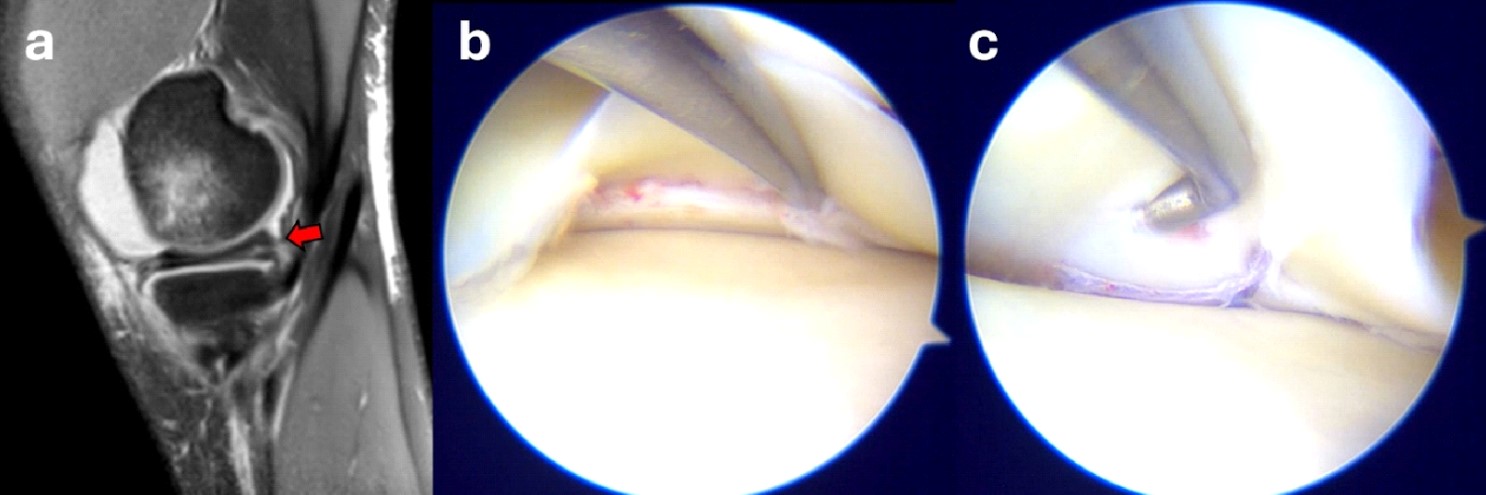
Figure 2. Sagittal MRI images showing a lesion of the medial meniscus posterior horn (red arrow) in combination with anterior tibial eminence fracture (a); arthroscopic view before (b) and after (c) all-inside meniscal repair.
Indications for ARIF
Generally, ARIF can be considered in all fractures where a surgical indication exists, thus in Type III and IV fractures of Meyers and McKeever’s classification. Borderline indication is in the Type II fracture, where the partial displacement can be reduced in full extension, thus making it suitable for conservative treatment. However, in the case of a Type II fracture that fails to reduce with closed methods or displays displacement greater than 3-5 mm, soft tissue interposition is possible, and thus, surgical reduction is necessary to restore joint congruence and prevent functional deficits. Surgical treatment of Type III and IV leads to a risk reduction of 56% in residual clinical knee instability (70% nonoperative vs. 14% operative, p < 0.0001), a risk reduction of 52.8% in patient-reported stability and of 9% in ACL reconstruction rate (10% vs. 1%, p = 0.036)16.
ARIF is also indicated when the fracture is associated with concomitant injuries such as meniscal tears or entrapment, ligamentous injuries (including the ACL), or cartilage damage. Studies17,18 have shown that up to 68.8% of patients with tibial spine fractures also suffer from associated soft tissue injuries, which makes a comprehensive arthroscopic evaluation essential during surgery. Arthroscopy allows the surgeon to address these additional injuries in a minimally invasive manner, which is another reason why ARIF is preferred over open techniques.
The choice of ARIF over open surgery is often driven by its advantages, which include a smaller surgical incision, reduced postoperative pain, lower risk of infection, and faster recovery times. Arthroscopic surgery also allows for better visualization of the fracture site and the ability to assess and treat associated intra-articular pathologies without the need for large incisions. In contrast, open reduction can increase the risk of scar formation, prolonged recovery, and higher complication rates, including joint stiffness13.
The analysis of the complications’ risk factors suggests that the surgical approach did not independently increase the risk of arthrofibrosis. ARIF technique may be indirectly exposed to other risk factors for arthrofibrosis because of the increased time between injury and surgery, waiting for a sports medicine surgeon, or because of a longer surgical time than in open procedures19. A prolonged surgical time is the strongest independent risk factor for arthrofibrosis when exceeding 120 minutes20.
Pre-Operative Planning
When the decision to perform ARIF in an anterior tibial eminence fracture is made, accurate preoperative planning should be performed. First of all, a precise diagnosis should be made: considering that the injury mechanism is similar to that of ligament injuries, collateral ligament and other intra-articular injuries should be identified, and their treatment included in the plan. All devices for meniscal and cartilage repair should be available during tibial spine ARIF, considering the possibility of unexpected injuries.
Then, the method of fixation should be chosen. Different techniques or devices, such as absorbable or nonabsorbable sutures, cannulated screws, bioabsorbable screws, K-wires, or metal cerclages, are available to obtain a solid and stable fixation, each with advantages and disadvantages.
The technique of choice for the senior author (A.G.) includes the use of transosseous repair with absorbable sutures, which allows for the treatment of most fractures (even comminuted ones) and does not require a second surgery for hardware removal. This method utilizes standard No. 2 absorbable sutures to secure the fragment by passing them through transosseous tunnels created in the tibia. The required instrumentation includes basic arthroscopic instrumentation, ACL guides to perform transtibial tunnels and suture passers to pass the stitches through the ACL substance.
Due attention needs to be paid to the fact that, if it is not possible to obtain an acceptable reduction arthroscopically, an open procedure could be necessary, and thus all the required instruments should be included in the preoperative setting.
Surgical Technique
Step 1: Patient Setup
The patient is positioned supine, with a tourniquet inflated. A lateral post is positioned 4 cm above the patella to allow the leg to hang free from the operating table to allow stress provocation. A fluid pump is routinely used. A medial suprapatellar portal is used for water inflow, while high anterolateral and standard anteromedial portals are used for the surgical instruments. Operating room setup should include space for a C-arm in order to perform intraoperative fluoroscopic radiographs if confirmation of fracture reduction is required. The C-arm should be positioned on the same side of the arthroscopic tower.
Step 2: Arthroscopic Assessment
The hemarthrosis is evacuated from the superomedial portal. Initially, it could be difficult to perform a careful intraoperative assessment due to hemarthrosis. If the water inflow through the superomedial portal causes blood dilution from the suprapatellar pouch, the inflow can be moved to the anterolateral portal.
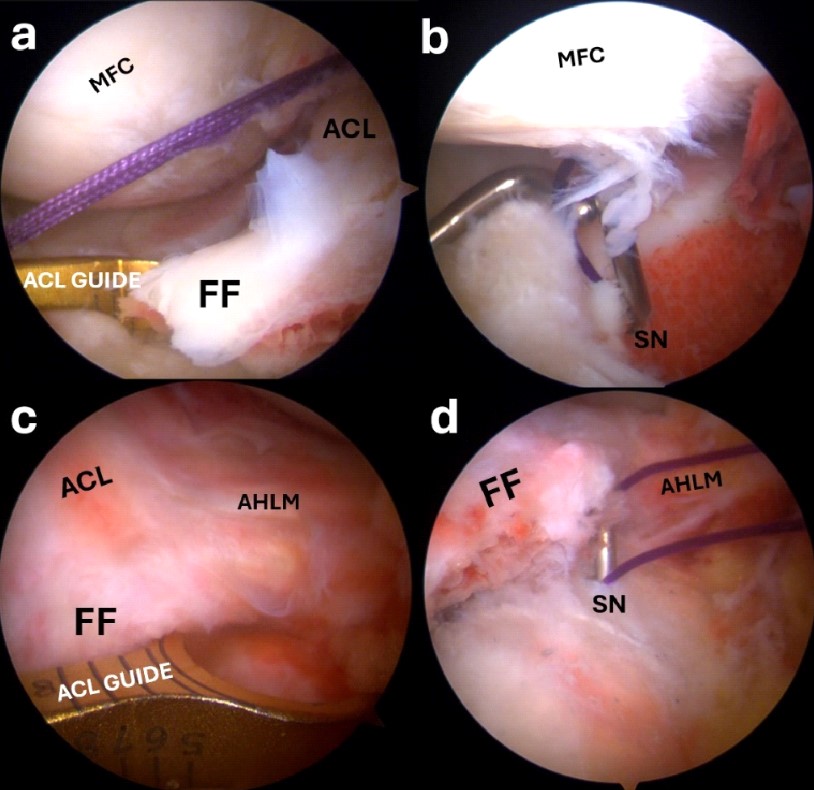
When enough visibility is obtained, the intraarticular hematoma is aspirated using a shaver through the anteromedial portal. During this stage, the ligamentum mucosum and part of the infrapatellar fat pad are resected to allow adequate visualization. After the hematoma has been evacuated, it is possible to inspect the joint for any other intra-articular lesions that should be addressed (Figure 2b, c). Care should be taken to check the condition and integrity of the anterior cruciate ligament (ACL) and the status of the articular cartilage and menisci, especially the anterior root of the lateral meniscus that could be avulsed with the spine. In this case, the anterior root of the lateral meniscus could displace the fracture fragment laterally and proximally (Figure 3). The fragment and its bony bed should be gently freed from hematoma and fibrous tissue to allow the study of the fracture pattern, keeping in mind the preoperative radiological exams. If the fracture is comminuted, small fragments (1-2 mm) should be removed to avoid producing loose bodies (Figure 4).
Figure 3. Coronal CT scan showing an anterior eminence fracture (red arrow) displaced laterally and proximally (a); the arthroscopic view shows connection of the anterior horn of the lateral meniscus with the main fracture fragment (b).
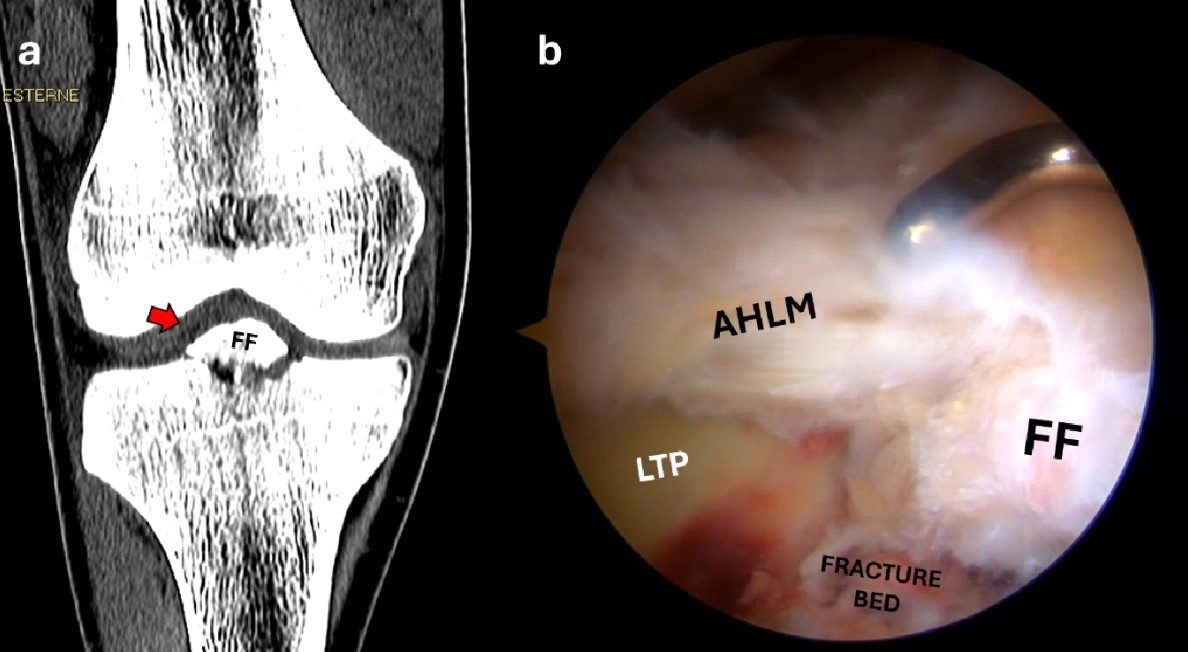
FF, fracture fragment; LTP, lateral tibial plateau; AHLM, anterior horn of the lateral meniscus.
Figure 4. Sagittal CT scan showing a displaced and multifragmentary anterior eminence fracture with a loose fragment (a); the arthroscopic view shows a small loose fragment which is removed with a grasper (b).
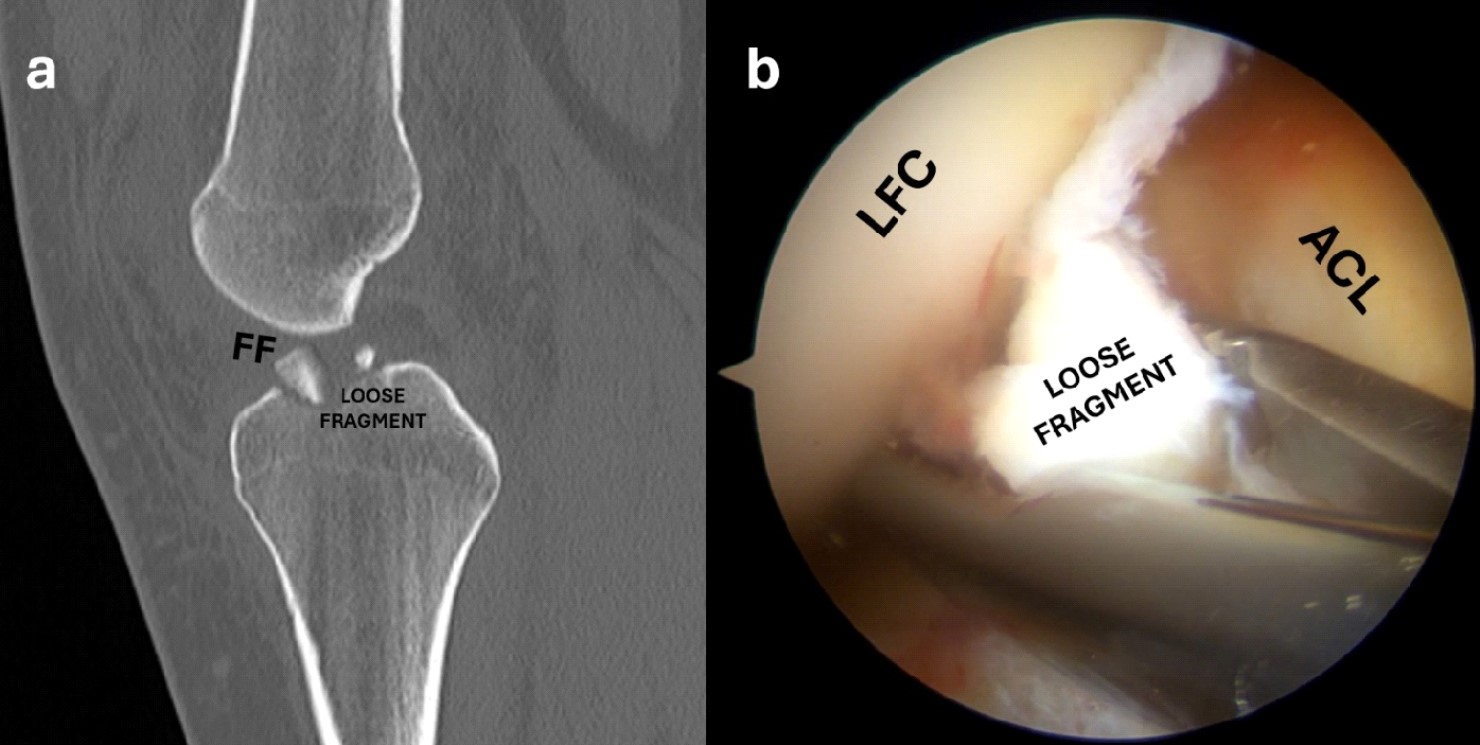
LFC, lateral femoral condyle; ACL, anterior cruciate ligament.
Step 3: Fragment(s) Reduction
Turning the optics anteriorly, the intermeniscal ligament is identified in order to check if any interposition is present with the fracture fragment. In such cases, the probe is introduced from the anteromedial portal to move the intermeniscal ligament, thus allowing reduction of the fragment. The intermeniscal ligament should be resected only in cases where it is impossible to disengage the entrapment (Figure 1). The fracture fragment is elevated, and the residual blood clot and debris are removed from the fracture bed with a shaver or small curette (Figure 5). In sub-acute cases, it is possible to find fibrous tissue in the anterior part of the fracture, making it impossible to elevate and mobilize the fragment. This should be carefully removed; otherwise, reduction may be impossible. Another aspect that requires careful consideration is the entrapment of the anterior horn of the medial meniscus between the fragment and the fracture bed (Figure 6); in this case, the probe can be used to retract the meniscus and allow fracture disengagement (Figure 7). Once the fragment is mobilized, the fracture can then be reduced into its bony bed using a probe inserted from the anteromedial portal. Extension of the knee can be helpful. Slight over-reduction of the fracture fragment is recommended.
Figure 5. Arthroscopic view after the cleaning of the fracture bed from hematoma and fibrous tissue; the fracture margins can be clearly seen.
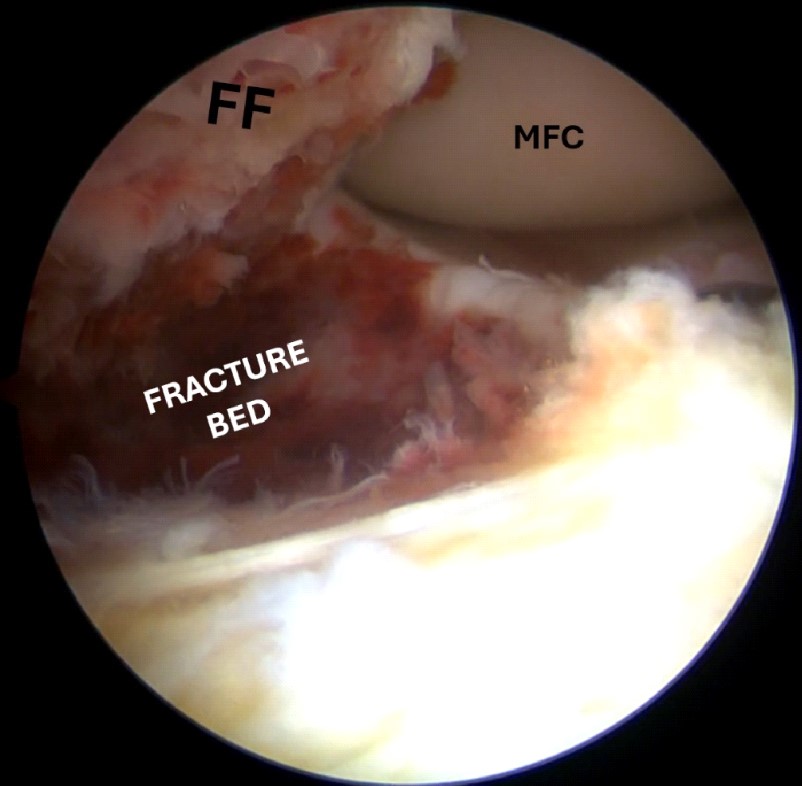
FF, fracture fragment; MFC, medial femoral condyle.
Figure 6. Sagittal CT scan showing a displaced anterior eminence fracture (red arrow) (a); the arthroscopic view shows the interposition of the anterior horn of the medial meniscus and the fracture bed (b).
FF, fracture fragment; MM, medial meniscus; AHMM, anterior horn of the medial meniscus; ACL, anterior cruciate ligament.
Figure 7. Arthroscopic view of medial meniscus entrapment in the fracture side; the anterior horn of the medial meniscus is retracted with the probe in order to disengage the fracture fragment.
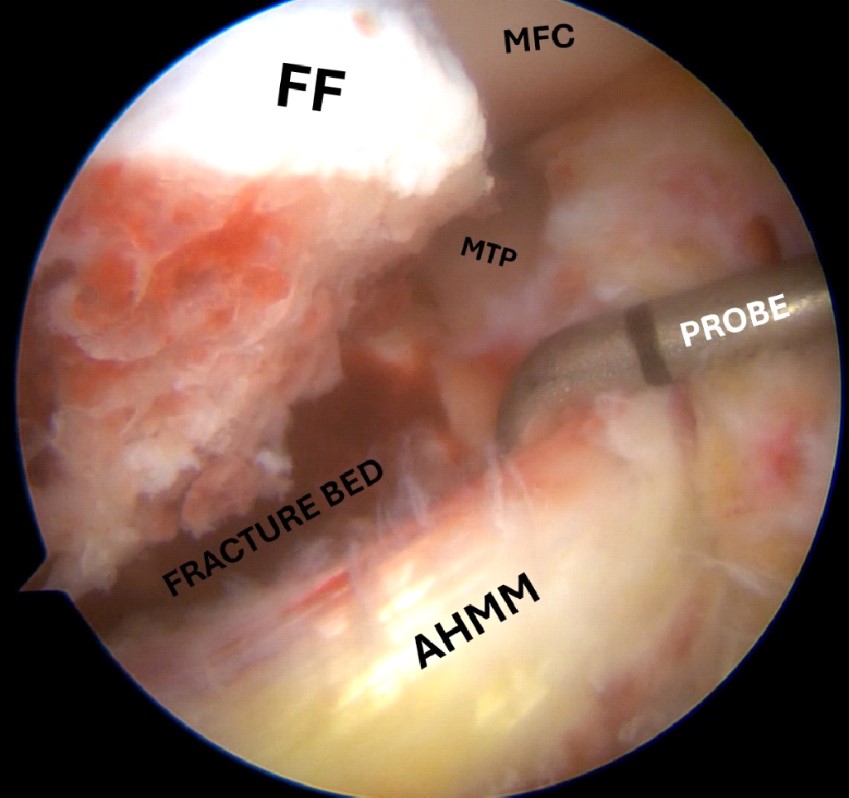
FF, fracture fragment; MTP, medial tibial plateau; AHMM, anterior horn of the medial meniscus.
Step 4: Preparation of Fixation
After the fragment reducibility is tested, a No. 2 absorbable Vicryl suture can be passed through the ligament using a suture passer (Figure 8a). Both ends of the suture are passed through the ACL substance in order to create a loop, which allows solid pulling and good grip (Figure 8b). Another No. 2 suture is passed after switching the portals to avoid confusing the two different pairs of sutures (Figure 8c). The first suture can be pulled in order to create a counter-tension when passing the second suture, if needed. However, pulling the suture too much does not guarantee fragment reduction and can rather break the posterior hinge (if any) and displace the fragment itself. To maintain the fracture reduced, without constantly pulling the sutures, a straight needle could be inserted within the ACL substance to temporarily fix it.
Now, a small tunnel is created with a 2.4 mm pin using an ACL guide, at the level of the anteromedial border of the fracture bed (Figure 9a), after creating a small skin incision in the anteromedial proximal tibia. During this maneuver, it could be necessary to gently elevate the intermeniscal ligament with an arthroscopic probe to ensure tunnel creation below it. A No. 1 PDS is passed inside a spinal needle to create a loop, and the needle is inserted inside the transosseous tunnel; the PDS loop is thus retrieved with the probe from the anteromedial portal (Figure 9b). Outside the joint, the two free ends of the Vicryl suture connected to the ACL are passed inside the PDS loop, which is pulled from the tibial side, retrieving the suture inside the tunnel. Since an arthroscopic cannula is not routinely used to maintain the portal, the two sutures cannot be passed simultaneously, resulting in soft tissue interposition during this maneuver. Hard pulling can break minor soft tissues; otherwise, a shaver should be used or the passage should be repeated.
The same steps are performed to create a second tunnel at the level of the anterolateral margin of the fracture bed, usually close to the anterior horn of the lateral meniscus (Figure 9c-d). A small cortical bridge should be left between the two tunnel entries on the anteromedial tibial aspect.
Figure 8. With the suture passer, an absorbable suture is passed through the ACL fibers (a). A loop around the ACL is created with the suture (b). Two loops are placed in the ACL from the two arthroscopic portals (c).
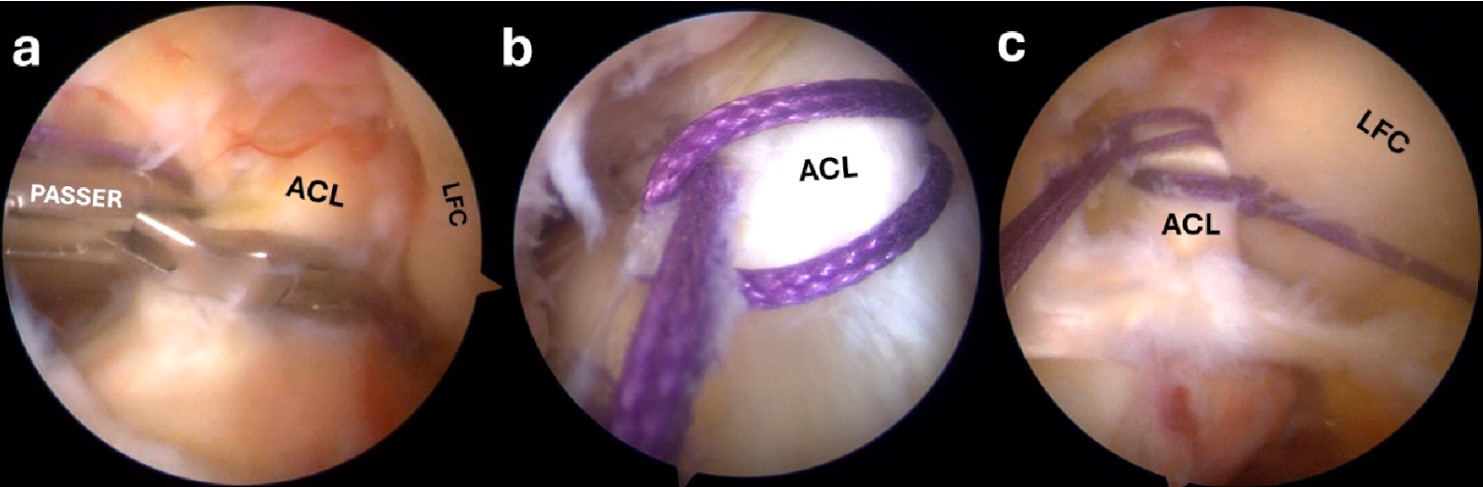
ACL, anterior cruciate ligament; LFC, lateral femoral condyle.
Figure 9. The ACL guide is positioned at the anteromedial margin of the fracture bed and a 2.4 mm tunnel is drilled (a); a spinal needle with a PDS loop is inserted in the tunnel and the suture retrieved with the probe (b); another tunnel is positioned after placing the ACL guide at the anterolateral margin of the fracture bed (c); another PDS suture is passed in the tunnel (d).
FF, fracture fragment; NS, spinal needle; ACL, anterior cruciate ligament; AHLM, anterior horn of the lateral meniscus.
Step 5: Final Fixation
When the two pairs of sutures are inside the two tunnels, a selective tension can be applied to each suture to reduce the fragment. It is not granted that pulling guarantees an “automatic” anatomical reduction; therefore, a grasper could be used to position the fragment in the proper position (Figure 10a). Once the reduction is satisfactory, the sutures could be firmly pulled by an assistant (Figure 10b); extending the knee could help in the reduction maneuver. Lack of impingement in full extension and the proper reduction of the margins (Figure 10c) should be verified before tying the two sutures on the cortical tibial bridge. Finally, stability during all ranges of motion and upon ACL probing should be confirmed.
Figure 10. A grasper is used to reduce the fracture fragment before pulling the sutures (a); when the desired reduction is obtained, the medial and lateral sutures are pulled simultaneously (b); the fracture lines (black arrows) are checked for any malreduction (c).
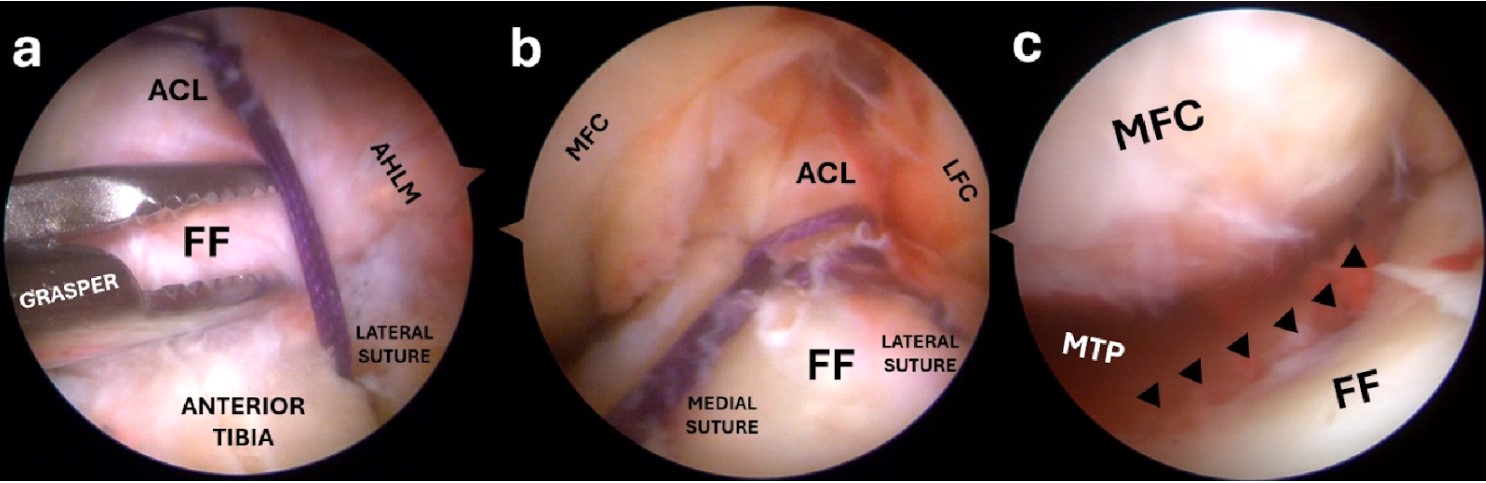
ACL, anterior cruciate ligament; FF, fracture fragment; AHLM, anterior horn of the lateral meniscus; MFC, medial femoral condyle; LCF, lateral femoral condyle; MTP, medial tibial plateau.
The “Plan B”: Conversion to ORIF
When it is impossible to reduce the fracture fragment during arthroscopy after more than 60 minutes of tourniquet time, we suggest switching to open reduction. A small anteromedial arthrotomy can be performed, extending the anteromedial arthroscopic portal. In this way, it is possible to access the fragment and fracture bed, which usually extends in a medial direction. It is also possible to disengage the interposition of the anterior horn of the medial meniscus, which is very close to the arthrotomy incision.
In open reduction and fixation, it is not necessary to change fixation methods; the ACL can be whipstitched under direct visualization, and the same transosseous suture technique could be used (Figure 11a-d). Alternatively, cannulated screws can also be used.
Figure 11. A Meyers and McKeevers Type IV fracture, preoperative anteroposterior (a) and lateral radiographs (c). Radiograph at 6-week follow-up after open reduction and internal fixation with transosseous sutures showing good reduction and healing of the fragment (b, d).
Postoperative Indications
The patient is allowed to start passive knee range of motion exercises, ranging from 0° to 90° in the first 2 weeks, and then progressively increasing. Partial weight-bearing is allowed on day 3 post-operatively with a knee brace locked in full extension. This gradually increases to full weight-bearing over the next 4 weeks. The patient is encouraged to return to routine activities of daily life by 8 weeks post-surgery. Patients can resume participating in non-contact sports within 3-4 months after surgery.
Discussion
Outcomes of ARIF
Clinical outcomes following ARIF are generally favorable, with a high rate of return to normal activity levels and sports participation. Multiple studies have documented positive results, with most patients achieving satisfactory functional recovery within 6 months. A study by Gans et al21 found that 85% of pediatric patients treated with ARIF for tibial spine fractures reported good to excellent outcomes according to the International Knee Documentation Committee (IKDC) scores. Another study22 comparing suture fixation vs. screw fixation found that suture-based techniques had a lower reoperation rate.
Suture techniques, in particular, have been shown to provide superior biomechanical properties compared to screw fixation, especially under cyclic loading15,23. This is due to the fact that suture fixation distributes the forces more evenly across the fracture site, reducing the risk of malreduction or failure under stress. Additionally, suture fixation eliminates the need for future surgeries to remove hardware, which is often necessary when screws are used.
Despite the positive outcomes, some complications can occur, including anterior knee pain, residual laxity, or knee stiffness24,25. Arthrofibrosis, or the formation of scar tissue leading to restricted range of motion, is the most common complication, occurring in up to 11% of cases. The most important risk factors for arthrofibrosis are prolonged immobilization, a delay of over 7 days until surgery, an operative time exceeding 120 minutes, and high-energy injuries with concomitant collateral ligament and meniscus injuries.
The use of early postoperative mobilization protocols has significantly reduced the incidence of this complication. Another potential issue is nonunion or malunion, which can occur if the fracture is not adequately reduced or if early weight-bearing is introduced before sufficient healing14. Residual laxity, in both surgical (10%) and non-surgical treatment (22%), is still a matter of concern. A key role in residual instability is due to interstitial damage to the ACL that occurred during the original injury. Moreover, 19% of all patients aged 5 to 18 years required subsequent ACL reconstruction, with the odds increasing by 1.3 for every year of increasing age at the time of tibial spine fracture26,27.
Illustrative Cases
In our clinical practice, ARIF has become the preferred treatment modality for anterior tibial spine fractures. The main factors guiding this choice are the typically young age of the patients, which prompts a preference for minimally invasive surgery, and the excellent direct visualization of the fracture site provided by arthroscopy.
ARIF allows accurate reduction and the simultaneous treatment of associated intra-articular lesions. ORIF is reserved only for those uncommon cases in which anatomical reduction cannot be achieved arthroscopically. The following cases represent emblematic examples of our ARIF experience, showcasing both typical and complex fracture patterns and their respective outcomes.
Case 1: A 17-year-old male sustained a right knee sprain during a fall while practicing motocross. The x-ray and CT showed a Meyers and McKeevers Type IV fracture (Figure 12a-b). After ARIF with absorbable sutures performed 5 days after injury, good reduction and healing were obtained at the 6-week follow-up evaluation (Figure 12c), where the patient demonstrated no antero-posterior (AP) laxity and complete range of motion (ROM). The patient returned to non-contact sports 3 months after injury.
Case 2: A 43-year-old male sustained a left knee sprain while skiing. The X-ray and CT showed a Meyers and McKeevers Type III fracture (Figure 13a-b). After ARIF with absorbable sutures performed 7 days after injury, good reduction and healing were obtained at the 6-week follow-up evaluation (Figure 13c), where the patient demonstrated no AP laxity and complete ROM. The patient returned to non-contact sports 3 months after injury.
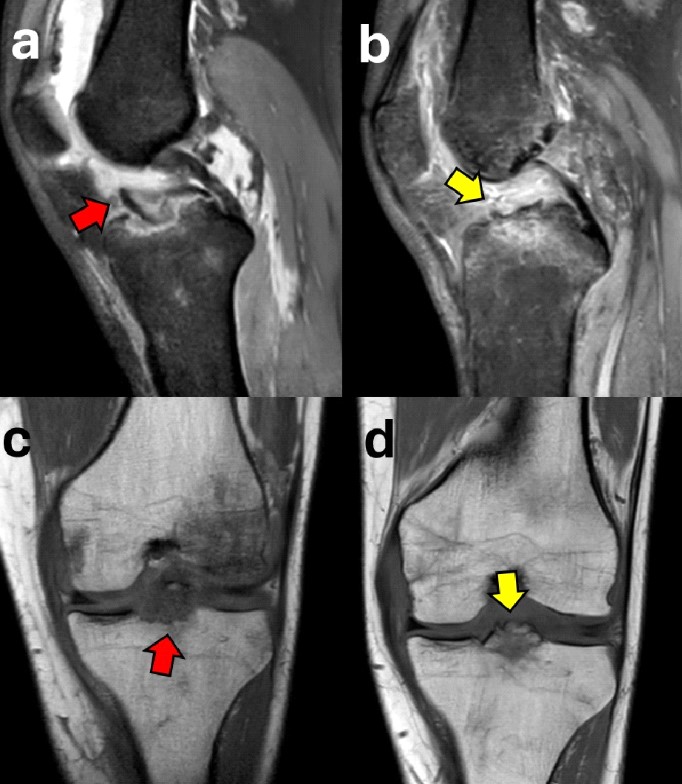
Case 3: A 24-year-old female tennis player sustained a left knee sprain while skiing. The initial X-ray was interpreted as negative, and the patient was initially treated conservatively in another institution. Due to the lack of extension, the patient underwent an MRI, which showed a Meyers and McKeevers Type III fracture (Figure 14a, c) with medial meniscus entrapment, medial meniscus posterior horn tear, and proximal medial collateral ligament (MCL) injury. After ARIF with absorbable sutures and medial meniscus repair performed 22 days after injury, good reduction and healing were obtained at the 8-week follow-up evaluation (Figure 14b, d). However, due to the late treatment, the patient developed arthrofibrosis, which was resolved after 6 months of intense rehabilitation. The patient returned to sport 7 months after injury.
Figure 12. A 17-year-old male with a Meyers and McKeevers Type IV fracture (red arrow) in sagittal CT scan (a) and lateral radiographs (b). Radiograph at 6-week follow-up showing good reduction and healing of the fragment (red arrow) (c).
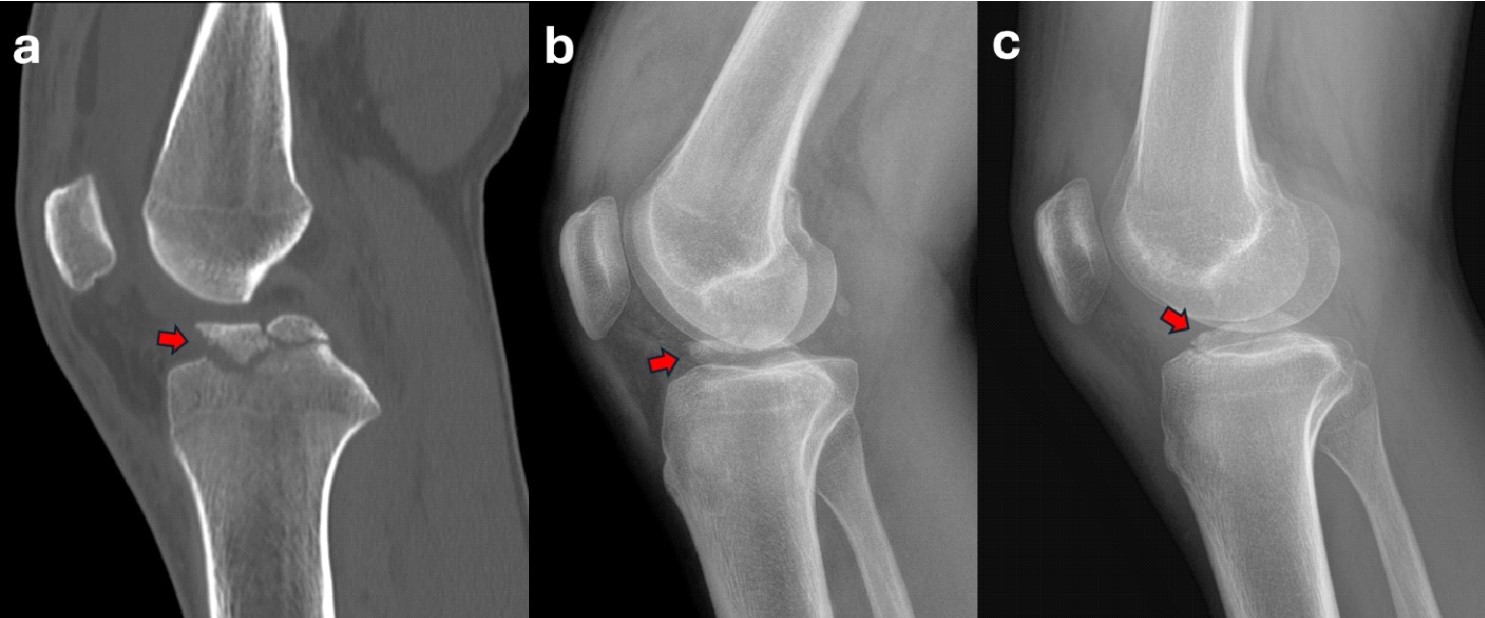
Figure 13. A 43-year-old male with a Meyers and McKeevers Type III fracture (red arrow) in sagittal CT scan (a) and lateral radiographs (b). Radiograph at 6-week follow-up showing good reduction and healing of the fragment (red arrow) (c).
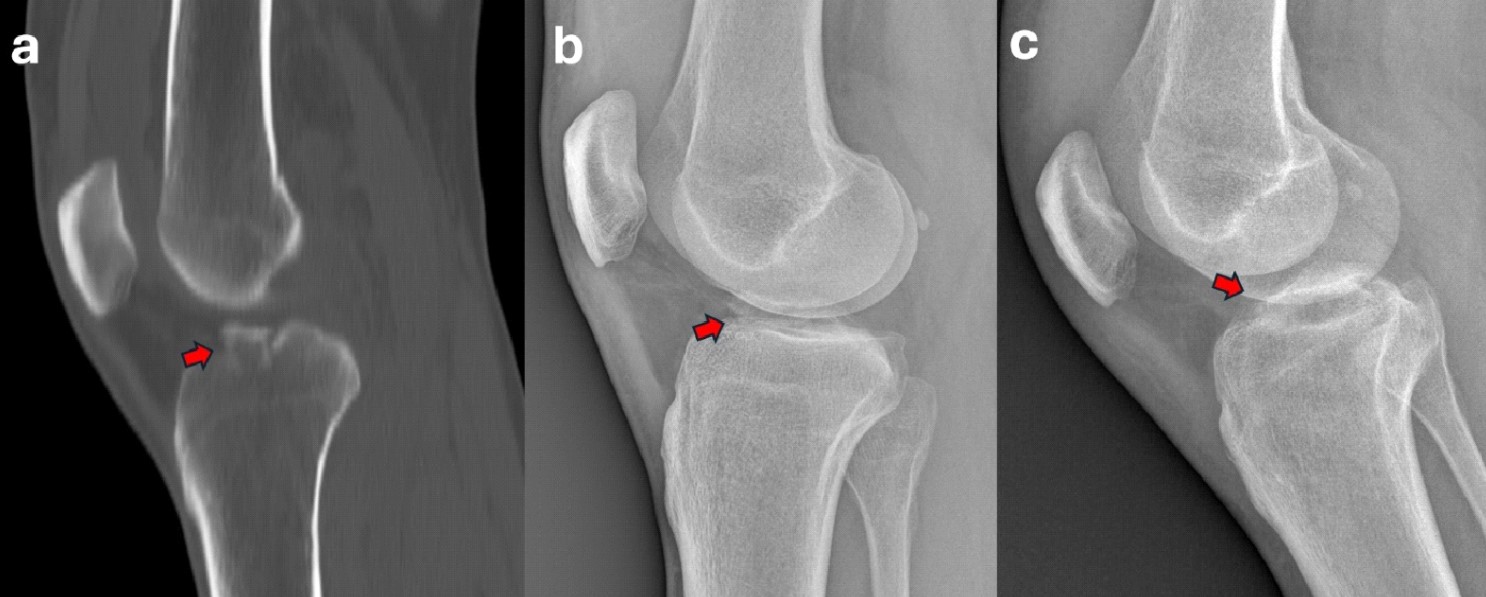
Figure 14. A 24-year-old female with a Meyers and McKeevers Type III fracture (red arrow) in a sagittal MRI scan (a) and the healed and reduced fragment (yellow arrow) 8 weeks after surgery (b); coronal pre-operative (c) and post-operative (d) views of the same patient.
Conclusions
Arthroscopic Reduction and Internal Fixation (ARIF) is an effective and minimally invasive surgical approach for the management of anterior tibial spine fractures. Its advantages are reduced postoperative pain, faster recovery and fewer complications compared to open surgery. Furthermore, arthroscopy also allows the treatment of associated soft tissue injuries that frequently complicate these fractures.
Transosseous suture fixation allows treatment of most fractures, ensuring biomechanical stability, and eliminates the necessity for future implant removal.
Prompt diagnosis, meticulous preoperative planning, and proper surgical management of these kinds of injuries are essential for a good outcome. ORIF remains necessary in certain complex fracture cases where satisfactory reduction is not achievable through arthroscopic techniques.
ORCID ID
Alberto Grassi: 0000-0003-4236-1798
Flavio Azzola: 0000-0001-6515-7533
Alberto Castelli: 0000-0002-8273-3923
Marcello Capella: 0000-0002-6076-8527
Funding
No funding was received.
Ethics Approval and Informed Consent
Ethical review and approval were waived for this technical note as it does not involve experimental intervention. Informed consent of the patients for the use of their data and images for scientific purposes was obtained when they agreed to be treated within the university hospital.
Conflict of Interest
The authors have no conflict of interest to disclose.
Authors’ Contributions
Alberto Grassi: conceptualization, writing – original draft.
Flavio Azzola: methodology, supervision.
Alberto Castelli: formal analysis, investigation.
Marcello Capella: data curation, writing – review and editing.
All authors read and approved the final version of the manuscript.
Data Availability
Supporting data will be made available by the authors upon reasonable request.
AI Disclosure
Artificial Intelligence (ChatGPT, OpenAI) was used to support abstract revision and language refinement.
References
1) DeFrancesco CJ, Tananbaum A, LeBrun DG, Fabricant PD. The Incidence of Pediatric Tibial Spine Fractures Is Greater and Peaks Later in Male Patients. Arthrosc Sports Med Rehabil 2022; 4: e639-e643.
2) DeFrancesco CJ, Wilson L, Lebrun DG, Memtsoudis SG, Fabricant PD. Pediatric Tibial Spine Fractures: Exploring Case Burden by Age and Sex. Orthop J Sports Med 2021; 9: 23259671211027237.
3) Perugia D, Basiglini L, Vadalà A, Ferretti A. Clinical and radiological results of arthroscopically treated tibial spine fractures in childhood. Int Orthop 2009; 33: 243-248.
4) Ellis HB, Zynda AJ, Cruz AI Jr, Sachleben B, Sargent C, Green D, Schmale G, Jagodzinski J, Rhodes J, Mistovich J, Fabricant P, McKay S, Lee R, Yen Y, Ganley T. Classification and Treatment of Pediatric Tibial Spine Fractures: Assessing Reliability Among a Tibial Spine Research Interest Group. J Pediatr Orthop 2021; 41: e20-e25.
5) Meyers MH, McKeever FM. Fracture of the intercondylar eminence of the tibia. J Bone Joint Surg Am 1959; 41-A: 209-222.
6) Green D, Tuca M, Luderowski E, Gausden E, Goodbody C, Konin G. A new, MRI-based classification system for tibial spine fractures changes clinical treatment recommendations when compared to Myers and Mckeever. Knee Surg Sports Traumatol Arthrosc 2019; 27: 86-92.
7) Kocher MS, Micheli LJ, Gerbino P, Hresko MT. Tibial Eminence Fractures in Children: Prevalence of Meniscal Entrapment. Am J Sports Med 2003; 31: 404-407.
8) Mitchell J, Sjostrom R, Mansour A, Irion B, Hotchkiss M, Terhune EB, Carry P, Stewart J, Vidal A, Rhodes J. Incidence of Meniscal Injury and Chondral Pathology in Anterior Tibial Spine Fractures of Children. J Pediatr Orthop 2015; 35: 130-135.
9) Shimberg JL, Leska TM, Cruz AI, Ellis HB, Patel NM, Yen Y, Tibial Spine Research Interest Group, Schmale GA, Mistovich J, Fabricant PD, Ganley JG, Green DW, Johnson B, Kushare I, Lee J, McKay SD, Milbrandt TA, Rhodes J, Sachleben B, Traver J. Is Nonoperative Treatment Appropriate for All Patients With Type 1 Tibial Spine Fractures? A Multicenter Study of the Tibial Spine Research Interest Group. Orthop J Sports Med 2022; 10: 23259671221099572.
10) Johnson AC, Wyatt JD, Treme G, Veitch AJ. Incidence of Associated Knee Injury in Pediatric Tibial Eminence Fractures. J Knee Surg 2014; 27: 215-219.
11) Wilfinger C, Castellani C, Raith J, Pilhatsch A, Höllwarth M, Weinberg AM. Nonoperative Treatment of Tibial Spine Fractures in Children-38 Patients With a Minimum Follow-up of 1 Year. J Orthop Trauma 2009; 23: 519-524.
12) Shin YW, Uppstrom TJ, Haskel J D, Green D W. The tibial eminence fracture in skeletally immature patients. Curr Opin Pediatr 2015; 27: 50-57.
13) Canfeng L, Xiancheng H, Qingjun Y, Yong L, Jiatong L, Sufen Y, Wenqian L, Xintao Z, Tian Y. Arthroscopic fixation techniques for tibial eminence fractures in pediatric patients: a review. Front Pediatr 2024; 12: 1347637.
14) Salvato D, Green DW, Accadbled F, Tuca M. Tibial spine fractures: State of the art. J ISAKOS 2023; 8: 404-411.
15) Eggers AK, Becker C, Weimann A, Herbort M, Thore Z, Raschke MJ, Petersen W. Biomechanical evaluation of different fixation methods for tibial eminence fractures. Am J Sports Med 2007; 35: 404-410.
16) Bogunovic L, Tarabichi M, Harris D, Wright R. Treatment of tibial eminence fractures: a systematic review. J Knee Surg 2015; 28: 255-262.
17) Tan Si Heng S, Fadzil K, Seng LAK, Hoipo HJ. Surgical management of tibial eminence avulsion fractures: a systematic review and meta-analysis. Arch Orthop Trauma Surg 2024; 144: 3153-3159.
18) Chandanani M, Jaibaji R, Jaibaji M, Volpin A. Tibial Spine Avulsion Fractures in Paediatric Patients: A Systematic Review and Meta-Analysis of Surgical Management. Children (Basel) 2024; 11: 345.
19) Smith HE, Cruz AI, Mistovich RJ, Leska TM, Ganley TJ, Aoyama JT, Ellis HB, Kushare I, Lee RJ, McKay SD, Milbrandt TA, Rhodes JT, Sachleben BC, Schmale GA, Patel NM. What Are the Causes and Consequences of Delayed Surgery for Pediatric Tibial Spine Fractures? A Multicenter Study. Orthop J Sports Med 2022; 10: 23259671221078333.
20) Watts CD, Larson AN, Milbrandt TA. Open versus arthroscopic reduction for Tibial eminence fracture fixation in children. J Pediatr Orthop 2016; 36: 437-439.
21) Gans I, Baldwin KD, Ganley TJ. Treatment and Management Outcomes of Tibial Eminence Fractures in Pediatric Patients: A Systematic Review. Am J Sports Med 2014; 42: 1743-1750.
22) Callanan M, Allen J, Flutie B, Tepolt F, Miller PE, Kramer D, Kocher MS. Suture Versus Screw Fixation of Tibial Spine Fractures in Children and Adolescents: A Comparative Study. Orthop J Sports Med 2019; 7: 2325967119881961.
23) Jain S, Modi P, Dayma RL, Mishra S. Clinical outcome of arthroscopic suture versus screw fixation in tibial avulsion of the anterior cruciate ligament in skeletally mature patients. J Orthop 2022; 35: 7-12.
24) Orellana KJ, Houlihan NV, Carter MV, Baghdadi S, Baldwin K, Stevens AC, Cruz Jr AI, Ellis HB, Green DW, Kushare I, Johnson B, Kerrigan A, Kirby JC, MacDonald JP, McKay SD, Milbrandt TA, Mistovich RJ, Parikh S, Patel N, Schmale G, Traver J, Yen YM, Ganley TJ. Tibial Spine Fractures in the Child and Adolescent Athlete: A Systematic Review and Meta-analysis. Am J Sports Med 2023; 52: 1357-1366.
25) Thaunat M, Barbosa NC, Gardon R, Tuteja S, Chatellard R, Fayard JM, Sonnery-Cottet B. Prevalence of knee stiffness after arthroscopic bone suture fixation of tibial spine avulsion fractures in adults. Orthop Traumatol Surg Res 2016; 102: 625-629.
26) Baums MH, Klinger HM, Härer T. Treatment of malunited fractures of the anterior tibial spine. Knee Surg Sports Traumatol Arthrosc 2004; 12: 159-161.
27) Mitchell JJ, Mayo MH, Axibal DP, Kasch AR, Fader RR, Chadayammuri V, Terhune EB, Georgopoulos G, Rhodes JT, Vidal AF. Delayed Anterior Cruciate Ligament Reconstruction in Young Patients With Previous Anterior Tibial Spine Fractures. Am J Sports Med 2016; 44: 2047-2056.
To cite this article
Arthroscopic reduction and internal fixation of anterior tibial spine: a practical guide
JOINTS 2025;
3: e1599
DOI: 10.26355/joints_20259_1599
Publication History
Submission date: 07 Jan 2025
Revised on: 07 Apr 2025
Accepted on: 09 Jul 2025
Published online: 15 Sep 2025